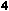Biodiversidade
https://www.sciencedirect.com/science/article/abs/pii/0006320794906122
M E A S U R I N G MORE OF BIODIVERSITY:
C A N H I G H E R - T A X O N RICHNESS PREDICT
WHOLESALE SPECIES RICHNESS?
P a u l H . W i l l i a m s a & K e v i n J. G a s t o n b
aBiogeography & Conservation Laboratory and bDepartment of Entomology, The Natural History Museum,
London, UK, SW7 5BD
(Received 26 June 1992; revised version received 27 April 1993; accepted 7 May 1993)
.tbstract
1"o assess conservation priorities, a means o f measuring the distribution of a much larger part o f overall bio- diversity is needed that will at the same time reduce the colossal sampling problems of exhaustive surveys. One possibility is a
Keywords: biodiversity, higher taxa, species richness, indicator.
INTRODUCTION
One of the problems conservationists face is how can the most important areas for biodiversity (biological diversity) be identified quickly and cheaply. Biodiversity is not only difficult to define, but basic
Maintenance of biodiversity has become one of the principal goals of conservation. Myers (1979) extended the existing concern for the more conspicuous and vulnerable species by drawing attention to the growing threat to the great variety of all living organisms.
Inevitably, it will not be possible to save everything
Biological Conservation
everywhere, so the conservation of biodiversity will have to be based on priority areas (McNeely et al., 1990; Groombridge, 1992; Reid et al., 1992). Many conflicting values need to be taken into account when assessing area priority (Usher, 1986; Spellerberg, 1992) and all should be made explicit and rational (Morowitz, 1991). Consequently we need to be able to measure the contribution an area makes to the overall pattern of biodiversity (May, 1990), preferably sum- marised in a simple univariate measure.
In practice, biodiversity is commonly measured by counting the number of species (species richness) in an area (Groombridge, 1992) and the turnover of species among areas
What is needed is a means of measuring the distribu- tion among areas of a much larger part of overall bio- diversity (Solbrig, 1991; Ehrlich, 1992), by which the sampling problem is reduced to manageable proportions (this larger part of overall biodiversity within an area is referred to here using the term wholesale biodiversity). Biodiversity surveys already take a large proportion of conservation budgets and the demand for them is growing:
Here we consider two familiar surrogates for esti- mating the distribution of overall biodiversity: environ- mental variables and indicator groups. A third approach, the use of
211

|
212 |
P. H. Williams, K. J. Gaston |
25
20
o
o
0
Fig. 1. Correlation coefficients calculated by Pianka and Schall (1981: Table 4) between species richness of groups of birds, reptiles, amphibians and mammals in Australia (161 of the 240 × 240 km grid squares), plotted as a frequency histogram. Comparisons are not independent, as some of the groups overlap in species composition: pairwise comparisons of marsupials, all birds, passerines,
varanids.
Biodiversity and environmental variables
Ecologists have searched for environmental factors that may limit overall biodiversity (e.g. Rabinovich & Rapoport, 1975; Schall & Pianka, 1978; Pianka & Schall, 1981; Wright, 1983; Currie & Paquin, 1987; Woodward, 1987; Owen, 1988; Turner et al., 1988; Adams, 1989; Adams & Woodward, 1989; Willig & Selcer, 1989; Currie, 1991). The use of characteristics of the physical environment as predictors of overall biodi- versity appears attractive because data for physical variables may already be available or may be relatively inexpensive to acquire. Some expensive verification, or
Unfortunately, many of the relationships that have been detected are severely
Biodiversity and indicator groups
Conservationists have sought to identify areas of high overall biodiversity by association with areas of rich- ness for relatively small, but particularly
relationships were shown to exist, sampling for just the selected species might greatly red..uce survey costs.
Birds, butterflies and most of'the other popular indi- cator groups have been surveyed particularly inten- sively over long periods because they have large body sizes and include representatives that are colourful and attractive (yet in all cases some taxa remain poorly known). But although they may be among the most obvious or conspicuous components of biodiversity, they are nonetheless only a very small proportion of overall biodiversity (Southwood, 1978; May, 1988; Barnes, 1989; Gaston, 1991a; Holloway & Stork, 1991; Hammond, 1992). Thus, use of these groups requires bold extrapolations, which need to be supported with good evidence for their reliability.
Unfortunately, areas of particularly high species rich- ness cannot be assumed to coincide among different groups of organisms. Even if some species' assemblages do show patterns of 'nested' composition at a local scale of distribution among habitat islands (Patterson
&Atmar, 1986; Cutler, 1991), this does not hold at the global scale. Lemurs are not indigenous to the New World and marmosets are not indigenous to the Old World, no matter how
Biodiversity and higher taxa
Here we consider another possibility, a
Intuitively, a relationship between diversity at differ- ent taxonomic levels appears likely. It is regarded as a good approximation in palaeontology (Sepkoski, 1992) and has been assumed for extant taxa (Salm, 1984). If a relationship can be demonstrated, then it might be possible to use the spatial distribution of

|
Biodiversity and higher taxa |
213 |
As an analogy, consider different numbers of objects (species) in a series of boxes (higher taxa), which are themselves present in different numbers among a series of rooms (localities). All that is being sought is whether or not there is a relationship between the distribution of boxes (higher taxa) among rooms (localities), and the overall distribution of objects (species) among those rooms (localities). Clearly it is essential to using the relationship that the level of effort to count boxes is not allowed to vary substantially among rooms, or else both the counts of boxes and of objects could simply reflect the relative level of effort expended in each room.
The key attraction of the
METHODS
We assessed the relationship between
Family richness and species richness for resident taxa were scored among all 100 x 100 km grid squares in Britain and Ireland, using published data for ferns and their allies (Perring & Walters, 1962), and for butter- flies (Heath et al., 1984). The 100 x 100 km
For the chosen organisms, families provide a reason- able compromise between sufficiently small numbers of higher taxa to be manageable and easy to survey, and sufficiently small average range sizes for the relation- ship to be informative (i.e. not all families are repre- sented in all areas). The taxonomic treatment accepted for each group follows that used in the sources of
distribution data, except for the butterflies. In this case we follow the more recent treatment of the Nymphalidae and Satyridae as a single family (R. I.
The relationships between the richness of species and higher taxa were tested using
RESULTS
All of the groups show significant positive correlations (p<0.001) between the numbers of families and the numbers of species in each grid square (untransformed data are plotted in Fig. 2). In every case, numbers of families account for >79% of the variance in the numbers of species. These correlations show that it is possible to use higher taxa as a surrogate for species in surveys of richness.
For the example data sets, family richness generally falls within two standard deviations of the mean rich- ness expected from choosing species at random (Fig. 2). The worst fit of the random model to the data is found for the butterflies, which show consistently fewer families in faunas than expected. This data set is the smallest among those studied, in terms of the number of families, and so is the least representative of the aims of the
DISCUSSION
The only properties required of higher taxa for estimat- ing wholesale biodiversity are (1) that their own richness distribution is predictive of the distribution of species richness, and (2) that information to map their distribu- tions is more readily acquired than for species. The first requirement is supported by our preliminary results.

214
P. H. Williams, K. J. Gaston
|
2 0 |
I |
|
I |
|
I |
I |
|
(a) |
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
i i i i i i i i i |
|
15 - - |
|
|
|
. . . ° |
~ I m t D ~ I M ~ , o |
|
|
|
|
|
|
o O ° ° ~ W |
~ o ~ o n |
, , ° ' ° |
|
|
|
j ml |
• | . ~ |
l i m m l l * |
||
|
|
|
mI |
i n a |
• |
l u I j |
|
|
|
• |
Oi n |
|
we l l j |
|
|
|
|
• |
mI |
0 . 1 |
n |
|
|
|
|
i i |
|
|
|
|
|
|
• |
I I |
l g |
|
|
|
|
|
. |
l i d i |
Is |
|
|
|
|
I
|
I I I |
i l l l l l |
° ' °~
. . o . ° "
_
6
5
4
i
3
|
. . |
I |
I |
I |
I |
I |
I |
|
~ |
|
|
•J* a l ° l l l u olmllll°elaamgulm llgme~ |
|||
|
- |
. . .. |
- |
" .............. |
......• - |
"1... |
|
|
~ O |
|
t ~ , , ~ l m W |
~ 0 |
|
|
|
|
|
i m |
|
|
|
• |
• |
|
mI |
|
|
|
|
|
• |
• |
|
|
|
|
• |
• |
|
|
|
|
|
|
|
|
|
|
|
i i I i I |
|
|
|
|
|
...o" |
|
|
|
|
|
i t |
|
|
|
|
i |
I |
|
|
|
|
0 |
I |
I |
I |
I |
|
10 |
20 |
30 |
40 |
|
|
|
|
|
S p e c i e s |
|
|
4O |
(c) |
u |
I |
I |
|
3O - - |
|
|
@ |
|
10
im
-@
|
I |
|
|
I |
I |
1 |
I |
I |
|
|
5 0 |
6 0 |
0 |
10 |
20 |
30 |
4 0 |
5 0 |
60 |
|
|
|
|
|
|
S p e c i e s |
|
|
|
|
|
|
10 |
(d) |
|
i |
|
|
|
|
|
|
|
|
|
|
|
8
•_
] 6 " m •
4
2
o
|
|
I |
I |
I |
|
|
l |
I |
|
0 |
50 |
100 |
150 |
200 |
0 |
50 |
100 |
|
|
|
S p e c i e s |
|
|
|
Species |
|
Fig. 2. Relationship between the numbers of families and numbers of species among areas (circles); (a) British ferns and their al- lies (100 × 100 km grid squares); (b) British butterflies (100 x 100 km grid squares); (c) Australian passerine birds, for l0° × l0° grid squares (O), and for alternate 5° × 5° grid squares ((3); and (d) North and Central American bats
Blakers et aL, 1984; and (d) Hall, 1981.
|
Some of the foreseeable problems and the second re- |
C o s t a d v a n t a g e s o f h i g h e r - t a x o n s u r v e y s |
|
quirement are discussed below. Further work is under- |
For practical surveys o f the diversity o f a broad range |
|
way to study the range of taxonomic and spatial scales |
o f organisms, determination o f the |
|
at which this relationship holds. We would expect that |
sition o f a fauna or flora should have the advantage of |
|
it would be most robust within the scope of cladistic |
being more |
|
revisions o f higher taxa by individual taxonomists. |
achieved than determination o f species richness. First, |
|
These are increasingly becoming available from studies |
there are obviously fewer higher taxa than species to |
|
at the scale o f major areas of endemism, biogeographic |
discriminate. Secondly, the highly uneven distribution |
|
'realms', continents, and the whole world. |
of species among higher taxa actually helps to increase |
|
If |
the rate at which samples can be sorted. The great |
|
wholesale species richness, then it could easily provide |
majority o f species belong to a small minority o f |
|
an overview of broad patterns in the distribution of bio- |
|
|
diversity from the wealth of existing |
Orchidaceae, and Asteraceae) and so can be quickly |
|
museums and libraries. Furthermore, the relationship |
dismissed. This 'hollow curve' frequency distribution o f |
|
should be applicable at finer spatial scales for practical |
lower taxa among higher taxa is a generally observed, |
|
conservation assessments o f local field surveys. |
although poorly understood, phenomenon (Willis, 1922; |

|
Biodiversity and higher taxa |
215 |
Williams, 1964; Clayton, 1972, 1974; Anderson, 1974; Bock & Farrand, 1980; Holman, 1985; Dial & Marzluff, 1989). It seems to hold both for species among higher taxa, and for other higher taxa among yet higher taxa. The distribution is likely to result in part from evolu- tionary processes (Wright, 1941; Anderson & Anderson, 1975; Glazier, 1987; Dial & Marzluff, 1989) and in part fi'om the way in which taxonomy is practised (Waiters, 1961, 1986; Clayton, 1972, 1974). So far, no model has provided a generally adequate description. Thirdly, species discrimination within many of the
Considerable resources could be saved by confining taxonomic effort to higher taxa. For example, with suitably prepared samples of tropical beetles, the over- all reduction in time and cost for a specialist counting family richness rather than species richness may be of the order of
Problem of uneven sampling effort
The problems of sampling for
In practice, one sampling strategy for higher taxa would be to try to effect an undirected search for in- dividual organisms. For example, certain collecting methods may be relatively 'blind' to higher taxa at some taxonomic ranks. An alternative sampling strategy would be to search in a manner directed specifically to the discovery of new higher taxa.
Problem of inappropriate rank of higher taxa
In order to preserve a close relationship with species richness for our present, pragmatic purposes, the choice of taxonomic rank at which to count higher taxa will
have to be made carefully. There has to be a compro- mise, because both the survey costs and the predictive value of the relationship should steadily decline at progressively higher taxonomic ranks. The strength of the relationship declines because an increasing propor- tion of the higher ranking taxa become widespread and uninformative, irrespective of spatial scale. Another possibility might be to combine taxa from different ranks (such as the
Problem of uneven taxonomic treatment
It is not essential for predicting species richness that higher taxa are either monophyletic (natural groups including all of the descendants of a common ancestor), or that they are necessarily of comparable rank in terms of age, or of phenetic or genetic distinctiveness. This is fortunate, because higher taxa of the same nomencla- tural rank are essentially arbitrary conventions, with no objective way of comparing the rank of taxa, except in the case of sister groups (Gauthier et al., 1988).
Another separate interpretation of higher taxa is that they represent higher level units of the difference or dis- parity among organisms (Williams et al., 1991). This interpretation is more demanding, because it really requires monophyly of higher taxa. However, it does allow information on genealogical relationships to be included in diversity measures to represent the degree of disparity among the organisms (Williams, 1993). Disparity has been seen as an important component of the concept of biodiversity (Gould, 1989; May, 1990;
The association between
Problem of atypical regions
Most biologists will be aware of regions of the world where there are genuine differences in the taxonomic richness relationship. Hawaii is a

|
216 |
P. H. Williams, K. J. Gaston |
also likely between the terrestrial and marine environ- ments, at least for the higher taxonomic ranks, because the marine fauna includes many more phyla but fewer described species (May, 1988; McNeely et al., 1990). Further studies are needed to determine whether atypical areas present a serious challenge, or whether they merely increase scatter about a general relationship.
Application to conserving more of overall biodiversity
The key to more representative estimates of overall bio- diversity is likely to lie with indirect approaches, such as those using environmental variables, indicator groups and
If suitable relationships between
ACKNOWLEDGEMENTS
Thanks to Josephine Camus, Paul Eggleton, Peter Hammond, Chris Humphries, Clive Moncrieff, Dick
REFERENCES
Adams, J.M. (1989). Species diversity and productivity of trees. Plants Today, 1989,
Adams, J.M. & Woodward, F.I. (1989). Patterns in tree species richness as a test of the glacial extinction hypothe- sis. Nature, Lond., 339,
Anderson, S. (1974). Patterns of faunal evolution. Q. Rev. Biol., 49,
Anderson, S. & Anderson, C.S. (1975). Three Monte Carlo models of faunal evolution. Amer. Mus. Novit., 2563,
Barnes, R.D. (1989). Diversity of organisms: how much do we know? Amer. Zool., 29,
Bibby, C.J. et al. (1992). Putting biodiversity on the map: priority areas for global conservation. ICBP, Cambridge.
Blakers, N., Davies, S.J.J.F. & Reilly, P.N. (1984). The atlas o f Australian birds. Melbourne University Press, Carlton.
Bock, W.J. & Farrand Jr, J. (1980). The number of species and genera of Recent birds: a contribution to comparative systematics. Amer. Mus. Novit., 2703,
Brown Jr, K.S. (1991). Conservation of Neotropical environ- ments: insects as indicators. In The conservation o f insects and their habitats, ed. N.M. Collins & J.A. Thomas. Academic Press, London, pp.
Burbidge, A.A. (1991). Cost constraints on surveys for nature
conservation. In Nature conservation: cost effective bio- logical surveys and data analysis, ed. C.R. Margules & M.P. Austin. CSIRO, Canberra, pp. 3 - 6 .
Clayton, W.D. (1972). Some aspects of the genus concept. Kew Bull., 27,
Clayton, W.D. (1974). The logarithmic distribution of angiosperm families. Kew Bull., 29,
Cronk, Q. (1988). Biodiversity. The key role of plants. IUCN and WWF, Gland.
Currie, D.J. (1991). Energy and
Currie, D.J. & Paquin, V. (1987).
Cutler, A. (1991). Nested faunas and extinction in fragmented habitats. Conserv. Biol., 5,
Dial, K.P. & Marzluff, J.M. (1989). Nonrandom diversifica- tion within taxonomic assemblages. Syst. Zool., 38,
Ehrlich, P.R. (1992). Population biology of checkerspot butterflies and the preservation of global biodiversity. Oikos, 63,
Gaston, K.J. (1991a). The magnitude of global insect species richness. Conserv. Biol., 5,
Gaston, K.J. (1991b). Body size and probability of descrip- tion: the beetle fauna of Britain. Ecol. Entomol., 16,
Gauthier, J., Kluge, A.G. & Rowe, T. (1988). Amniote phylogeny and the importance of fossils. Cladistics, 4,
Gentry, A.H. (1992). Tropical forest biodiversity: distribution patterns and their conservational significance. Oikos, 63,
Glazier, D.S. (1987). Energetics and taxonomic patterns of species diversity. Syst. Zool., 36,
Gould, S.J. (1989). Wonderful life. Penguin Books, London. Grinnell, J. (1922). The role of the 'accidental'. The Auk, 39,
Groombridge, B. (ed.)(1992). Global biodiversity. Status o f the Earth's living resources. Chapman and Hall, London.
Hall, E.R. (1981). The mammals o f North America, Vol. 1, 2nd edn. Wiley, New York.
Hammond, P.M. (1990). Insect abundance and diversity in the
Hammond, P.M. (1992). Species inventory. In Global biodi- versity status o f the Earth's living resources, ed. B. Groom- bridge. Chapman and Hall, London, pp.
Heath, J., Pollard, E. & Thomas, J. (1984). Atlas o f butterflies in Britain and Ireland. Viking, Harmondsworth.
Holloway, J.D. & Stork, N.E. (1991). The dimensions of bio- diversity: the use of invertebrates as indicators of human impact. In The biodiversity o f microorganisms and inverte- brates: its role in sustainable agriculture, ed. D.L. Hawksworth. CABI, Wallingford,
Holman, E.W. (1985). Evolutionary and psychological effects in
Kremen, C. (1992). Butterflies as ecological and biodiversity indicators. Wings, 16,
Landres, P.B., Verner, J. & Thomas, J.W. (1988). Ecological uses of vertebrate indicator species: a critique. Conserv. Biol., 2,
Loope, L.L., Hamann, O. & Stone, C.P. (1988). Comparative conservation biology of oceanic archipelagos. BioSci., 38,
MacKinnon, J. & MacKinnon, K. (1986). Review o f the protected areas system in the afrotropical realm. IUCN and UNEP, Cambridge.
May, R.M. (1988). How many species are there on Earth? Science, N. Y., 241,
May, R.M. (1990). Taxonomy as destiny. Nature, Lond., 347,

|
Biodiversity a n d higher taxa |
217 |